|
 Brachial Plexus Forum
Brachial Plexus Forum
Share ideas, lifestyle,
techniques interesting
observations and any other
issues you wish to discuss
|
|
|
|
|
Surgery
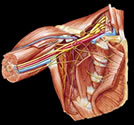 At
surgery, patients were placed in the lawn- chair position,
with a transverse shoulder roll. The head and neck were abducted
away from the side of surgery. The entire supraclavicular area was
prepared and draped with a thyroid sheet. The skin incision was
created one fingerbreadth posterior and parallel to the clavicle.
The incision was sinusoidal and extended 6 to 8 cm. lateral to the
palpated lateral clavicular border of the sternocleidomastoid muscle.
Dissection proceeded through the platysma muscle, taking care to
protect the underlying supraclavicular nerves. The omohyoid muscle
was resected to allow access to the scalene fat pad and to remove
a potential compressive structure of the brachial plexus. The scalene
fat pad was elevated from inferior to superior, revealing the upper
brachial plexus. Great care must be taken to identify the suprascapular
branch of the upper trunk, as it tends to travel within the middle
layers of the scalene fat pad, and is theoretically prone to iatrogenic
injury at this point. At
surgery, patients were placed in the lawn- chair position,
with a transverse shoulder roll. The head and neck were abducted
away from the side of surgery. The entire supraclavicular area was
prepared and draped with a thyroid sheet. The skin incision was
created one fingerbreadth posterior and parallel to the clavicle.
The incision was sinusoidal and extended 6 to 8 cm. lateral to the
palpated lateral clavicular border of the sternocleidomastoid muscle.
Dissection proceeded through the platysma muscle, taking care to
protect the underlying supraclavicular nerves. The omohyoid muscle
was resected to allow access to the scalene fat pad and to remove
a potential compressive structure of the brachial plexus. The scalene
fat pad was elevated from inferior to superior, revealing the upper
brachial plexus. Great care must be taken to identify the suprascapular
branch of the upper trunk, as it tends to travel within the middle
layers of the scalene fat pad, and is theoretically prone to iatrogenic
injury at this point.
Once the scalene
fat pad was elevated, the upper trunk and its trifurcation
into the anterior and posterior divisions and the suprascapular
nerve was explored. Typically, epineurial scarring was evident at
this point, and external neurolysis with microsurgical instruments
and technique was performed. Anterior scalene resection was also
performed at this time, although this was generally partial, and
only sufficient to release the most superficial fibers compressing
the upper trunk. This typically amounted to 15 or 20% of the thickness
of the anterior scalene muscle.
The long thoracic
nerve was then exposed laterally and posteriorly to the upper trunk.
Disa and colleagues point out the underappreciated anatomy of the
long thoracic nerve in the supraclavicular area, and discuss the
inadequacy of standard gross anatomic descriptions showing the nerve
to be more medial. It is worth noting that the long thoracic nerve
is delicate and no more than 2 to 3 millimeters in diameter in this
location. Its lack of substance in relation to the bulk of the serratus
anterior muscle teleologically predisposes the neuromuscular unit
to dysfunction. The situation is made worse by the passage of the
nerve through the thick middle scalene muscle.
Once the nerve was
isolated, it was then neurolysed using microsurgical instruments
and the operating microscope. This was necessary because of the
delicate nature of the nerve and to decrease surgical scar formation
within the operative field. As with the anterior scalenectomy, the
middle scalene was resected sufficiently to decompress the long
thoracic nerve as it traversed and exited the muscle. In 6 of 22
surgical procedures (27%), a demarcated area of compression within
the nerve was clearly noted, more so toward the exit point of the
nerve from the muscle. This took the form of a narrowing and rubor
of the epineurium, perhaps representing neovascularization at the
site of compression. In one case, a complete resection of the middle
scalene was required, and in the other 21 cases, a partial release
of 15% or slightly more was accomplished. This was suitable to expose
the long thoracic nerve and at least remove the circumferential
muscle fibers of the middle scalene.
Direct electrical
stimulation of the long thoracic nerve and upper trunk was performed
in all cases, with a Radionics ™ intraoperative nerve
stimulator. Current of up to 10 milliamps was used to stimulate
contraction of the serratus anterior and the muscles supplied by
the upper trunk. It was interesting that the contractions of the
serratus anterior appeared uniformly to improve following decompression
and neurolysis. It is a point of speculation as to the importance
of this electrical "overstimulation" of the nerves in
assisting recovery of the paralyzed muscles. It is compelling to
note that even in cases where no clear anatomic point of epineurial
compression could be noted, recovery of serratus function was excellent.
Further, in fully 50% of cases, recovery was noted within 24 hours,
an unusual circumstance in most situations of nerve decompression.
Prior to closure,
an examination of the superior- most and inferior margins
of the surgical wound was performed to identify and release compressive
fascial bands potentially capable of causing compression of the
upper trunk and the long thoracic nerve. This was accomplished sharply
under direct vision and with blunt digital dissection into the recesses
of the wound.
Wounds were closed in three layers
with reconstruction of the platysma and two skin layers.
No drains were used. Postoperative management consisted of immediate
active range of motion at the shoulder and neck. By the third postoperative
day, patients were to have a full range of motion at preoperative
levels or beyond, where capable.
|